 Supplements 1, 2, and 3 of the Journal of the Physical Society. There are fourteen types of lattices possible in a crystal. Crystal Lattice Defects: Proceedings of the International Conference on Crystal Lattice Defects. Q: How many kinds of space lattices are possible in a crystal?Īns: The correct option is “A”. End Centered: Has particles at the corners and one particle at the center of the opposite faces.Face Centered: This contains particles on every face of the lattice and other particles on the corners.Other particles (one or more) are at the corner of the lattice Body Centered: It has one particle at the center of the body.There are actually three types of non-primitive unit cells, namely: 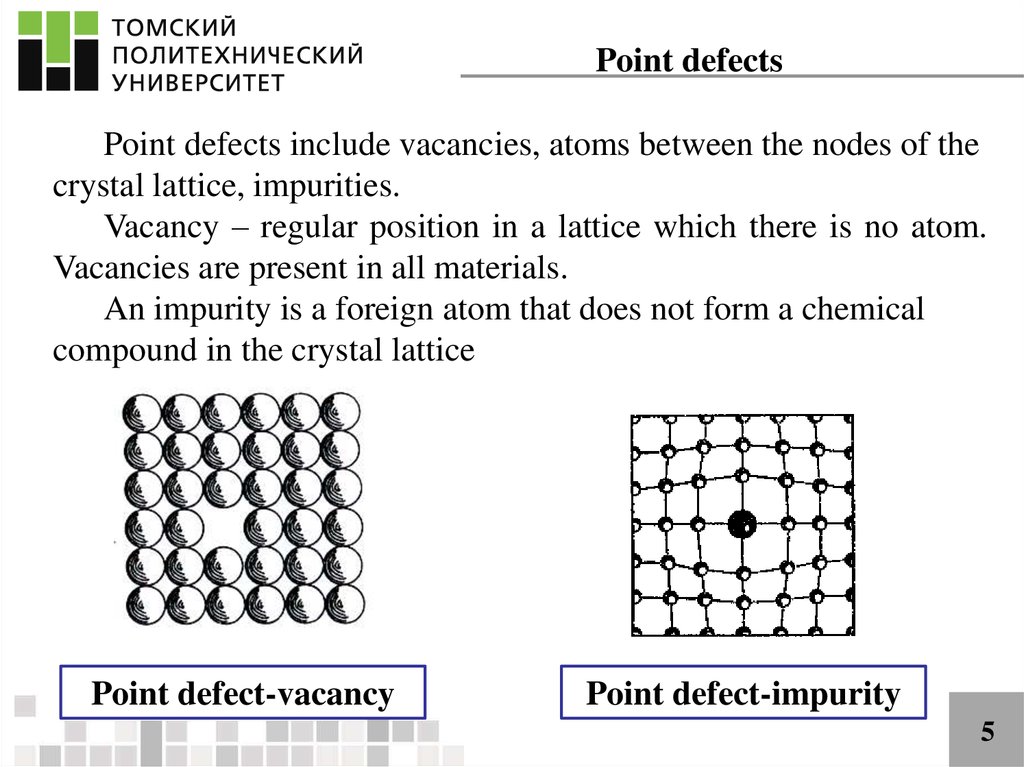
So there is more than one lattice point in a non-primitive unit cell. These additional constituent particles are either on the face of the unit cell or inside the unit cell. In this type of unit cell, there are particles not only at the corners of the lattice but in other positions as well. So essentially primitive unit cell has only one lattice point. There are no particles located at any other position in a primitive unit cell. The interfacial angles of the unit cell are as follows:Ī primitive unit cell only has atoms, molecules or ions at the corners of the lattice. C: edges defined by lattice vectors a and b. 
B: edges defined by lattice vectors a and c.A: edges defined by lattice vectors b and c.And these three edges form three respective angles. A unit cell is a geometric shape even by itself. The entire of the space lattice is built by the repeating arrangement of unit cells. It is used to visually simplify the crystalline patterns solids arrange themselves in. Every one of the fourteen lattices has such a unique geometryĪ unit cell is the most basic and least volume consuming repeating structure of any solid.By joining of these points we get the geometry (or shape) of the crystal.These lattice points of a crystal are joined together by straight lines.This particular particle may be an atom, a molecule or even ions.Each point on the lattice represents one particle of the crystal, This is a lattice point.It is the simplest repeating unit in a crystal structure. The fourteen Bravais Lattices show some similar characteristics. Unit Cell is the smallest part (portion) of a crystal lattice. The following diagram shows you the fourteen arrangements. The latter crystallises Bhagavantam and Pantulu. These 14 arrangements are the Bravais Lattice. 36 Defects in Crystal Lattices f Current L Science Calcium oxide (CaO) goes into solid solution loped in some detail by Wachtman4 and by in thorium oxide (Th02). 
Lattice structures (or crystals) are more prone to defects when their temperature is greater than 0 K. Now Auguste Bravais was French scientist who found out that there are a total of fourteen possible three-dimensional lattices. However, the arrangement of atoms or molecules in most crystalline materials is not perfect and the regular patterns are interrupted by crystallographic defects. So a lattice is an array of points in a particular order which describes the arrangement of particles of a crystalline solid. When such an arrangement of atoms is represented in a three-dimensional structure, this is a crystal lattice. Let us take a look.Ĭrystals have a structure made up of a regular arrangement of their atoms (or particles). Another is a crystalline structure or crystals which have a specific organized structure of their particles. One is an amorphous solid which has no specific shape or structure. During deformation, progressive movement along flaws in crystals takes place in tiny steps, as bonds are broken and re-formed.As we have studied in the previous topic, solids are basically of two shapes. Such defects affect the mechanical strength of a crystal, which determines the strength of rocks and how they deform under intense pressure. Figure 22 shows an artificial example based on an alloy used in semiconductors, where each dark line represents a strained part of the crystal. 
Minerals in deformed rocks, such as those from mountain belts, contain large numbers of line defects caused by rows of atoms that are out of place in the crystal structure. This is important because the rate of diffusion of atoms through a crystal structure can determine the speed at which processes such as weathering, or other chemical reactions (e.g. Such defects make it much easier for atoms to diffuse through the crystal structure by moving between vacant sites. Point defects may involve missing or displaced atoms in the crystal structure, giving empty sites, or vacancies. The effect of defects on the physical and chemical properties of a crystal can be out of all proportion to their size.ĭefects come in several types. Virtually all crystals contain minute imperfections or defects. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
